Vasopressin Analogs
General Pharmacology
Vasopressin (arginine vasopressin, AVP; antidiuretic hormone, ADH) is a nonapeptide hormone formed in the hypothalamus and released from the posterior pituitary. Its primary function in the body is to regulate extracellular fluid volume by affecting renal handling of water; however, it also is a potent vasoconstrictor.
There are several mechanisms regulating the release of AVP. Hypovolemia, as occurs during hemorrhage, results in a decrease in atrial pressure. Specialized stretch receptors within the atrial walls and large veins entering the atria decrease their firing rate when there is a fall in atrial pressure. Afferent nerve fibers from these receptors synapse within the nucleus tractus solitarius of the medulla, which sends fibers to the hypothalamus, a region of the brain that controls AVP release by the pituitary. Atrial receptor firing normally inhibits the release of AVP by the posterior pituitary. With hypovolemia or decreased central venous pressure, the decreased firing of atrial stretch receptors leads to an increase in AVP release. Hypothalamic osmoreceptors sense extracellular osmolarity and stimulate AVP release when osmolarity rises, as occurs with dehydration. Finally, angiotensin II receptors in a region of the hypothalamus regulate AVP release – an increase in angiotensin II simulates AVP release.
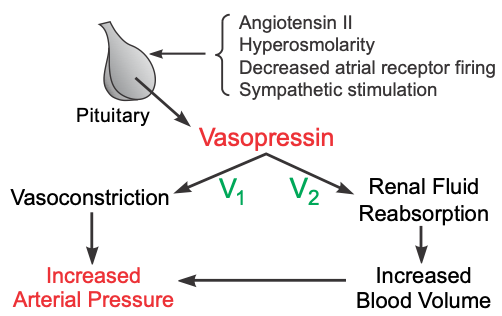 AVP has two principal sites of action: the kidney and blood vessels. The most important physiological action of AVP is to increase water reabsorption in the kidneys by increasing water permeability in the collecting duct, permitting the formation of more concentrated urine. This is the antidiuretic effect of AVP, and it acts through vasopressin type 2 (V2) receptors coupled to adenylyl cyclase. AVP also constricts arterial blood vessels by binding to V1 receptors, which are coupled to the Gq-protein and the IP3 signal transduction pathway. The Rho-kinase pathway is also activated and contributes to the smooth muscle contraction. Normal physiological concentrations of AVP are below its vasoactive range; however, in hypovolemic shock when AVP release is very high, AVP contributes to the compensatory increase in systemic vascular resistance.
AVP has two principal sites of action: the kidney and blood vessels. The most important physiological action of AVP is to increase water reabsorption in the kidneys by increasing water permeability in the collecting duct, permitting the formation of more concentrated urine. This is the antidiuretic effect of AVP, and it acts through vasopressin type 2 (V2) receptors coupled to adenylyl cyclase. AVP also constricts arterial blood vessels by binding to V1 receptors, which are coupled to the Gq-protein and the IP3 signal transduction pathway. The Rho-kinase pathway is also activated and contributes to the smooth muscle contraction. Normal physiological concentrations of AVP are below its vasoactive range; however, in hypovolemic shock when AVP release is very high, AVP contributes to the compensatory increase in systemic vascular resistance.
Specific Drugs
Arginine vasopressin (AVP) is used in the treatment of patients in shock. Terlipressin (triglycyl lysine vasopressin) is a long-acting vasopressin analog that is under clinical investigation. In contrast to AVP, this analog has a relatively higher affinity for vascular V1 receptors than for renal V2 receptors.
Therapeutic Uses
The main uses of AVP are for treating excessive water loss caused by diabetes insipidus, for treating bleeding caused by esophageal varices, and as a pressor agent in treating septic shock, which is a vasodilated, hypotensive condition that can be caused by infection and inflammation. Infusion of AVP in septic shock increases systemic vascular resistance and elevates arterial pressure. AVP should be considered when fluids and other vasopressor agents (e.g., vasoconstrictor catecholamines) cannot restore arterial pressure to an adequate level. Some studies have shown that low-dose infusions of AVP (which are used in septic shock) also cause cerebral, pulmonary and renal dilation (mediated by endothelial release of nitric oxide). The overall effect is an increase in systemic vascular resistance. AVP is also being investigated for other forms of shock, such as cardiogenic and hypovolemic (hemorrhagic) shock, but its benefit is less clear than for septic shock.
Side Effects and Contraindications
Side effects include headache, nausea, bronchoconstriction and abdominal cramps. Its antidiuretic effects can lead to water intoxication and hyponatremia. Because of AVP's powerful constrictor response, it should be administered cautiously to patients with coronary artery disease because it constricts coronary arteries (reducing oxygen delivery) and increases myocardial oxygen demand by increasing afterload on the heart.
Revised 11/30/2023
 Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021)
Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021) Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)
Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)